The procedure of single-port inflatable mediastinoscopy and laparoscopic surgery for radical esophagectomy
Introduction
Radical esophagectomy is very important for improving the overall survival and quality of life in patients with esophageal cancer (1,2). Traditional esophagectomy includes transthoracic and trans-hiatal approach (3,4). Transthoracic approaches have open and minimally invasive esophagectomy (MIE), such as Swee, Ivor-Lewis, and McKeown approach (1). Potential advantages of MIE include: the lower cardiopulmonary complications, less post-operative wound infection, decreased post-operative pain, and shorter length of hospitalization than conventional open esophagectomy (5,6). Robotic esophageal surgery is a type of transthoracic MIE surgery and still has disadvantages of single lung ventilation in surgery and many postoperative cardiopulmonary complications (7). Transhiatal esophagectomy is regarded as a type of MIE because of avoiding open thoracotomy, but it is still considered less accurate because it only shows a minimal surgical view and deficient mediastinal lymphadenectomy by conventional specialized mediastinoscopy (8). Recently some clinical studies on non-transthoracic radical esophagectomy have been reported from Japan and China (9). This report focuses on the novel minimally invasive techniques for esophageal cancer surgery, which is single-port inflatable mediastinoscopy and laparoscopic surgery (10). The procedure is described in detail.
Preoperative preparation
The pathological diagnosis of esophageal cancer should be clarified preoperationally by gastroscopy biopsy, surgical indications of the patients should be evaluated, and medical contraindications should be excluded. The preoperative tests included Blood routine, liver and kidney function, coagulation function related to surgery, electrocardiogram, echocardiography, neck/chest and abdomen enhanced CT, ultrasound gastroscopy, PET-CT, pulmonary function test, upper gastrointestinal angiography.
Anesthesia
The procedure is performed under general anesthesia. The central venous catheter should be placed in the right subclavian vein. A single-lumen endotracheal tube is used for double-lung ventilation. Due to the intraoperative use of carbon dioxide gas source to produce artificial mediastinal emphysema and pneumoperitoneum, blood gas analysis should be performed every 30 molecules to prevent the accumulation of carbon dioxide. Accumulated blood carbon dioxide can be regulated by raising the ventilation frequency or suspended carbon dioxide gas source.
Patient position and surgeon position
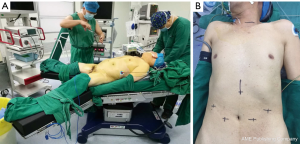
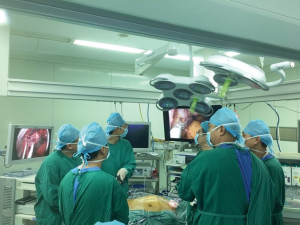
The patient lies in the supine position with the legs apart. The operating table leans to the right, and the left shoulder is raised 10 cm with a soft pad. This position makes it easier for the surgeon to perform mediastinal and intraperitoneal operations (Figure 1). In the mediastinal operating section, the surgeon stands on the patient’s cephalic side, the photographer is on the surgeon’s right side, and the scrub nurse is on the patient’s left side. In the abdominal operating section, the surgeon stands between the patient's legs. The assistant stands on the right of the patient and the photographer and scrub nurse on the left (Figure 2).
Surgical procedure
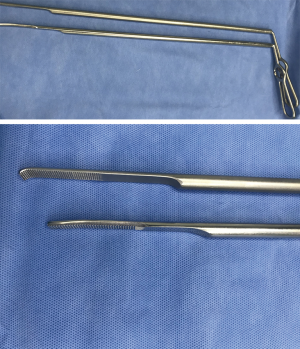
The upper mediastinal retractor (35 cm, Ø5 mm) (Figure 3) and the lower mediastinal retractor (45 cm, Ø10 mm) (Figure 4) (manufactured by Suzhou Sagemed Medical Technology Co., Ltd. China) and the Maryland forceps (model: endoscopic 1737, Medtronic, Covidien) were the essential tools for this novel non-trans thoracic esophagectomy.
The procedure mainly consisted of two parts: upper mediastinal operation and laparoscopic operation. Figure 2 showed the positions of the operator and assistants stood in the upper mediastinal procedure.
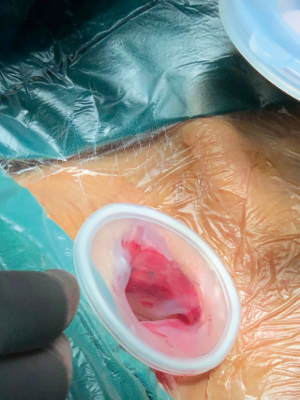
For upper mediastinal operation, lateral incision of the clavicle was made about 3 cm from the supraclavicular region in the left neck. Through the incision, anterior cervical muscles were cut with an electric scalpel, and the left internal jugular vein and the left common carotid artery were exposed by pulling the inside margin of the left sternocleidomastoid muscle outward. Then the cervical esophagus was exposed by pulling them with a hook. The cervical lymph nodes along the left recurrent laryngeal nerve (RLN) were removed and the RLN clarified through the inferior thyroid gland. The cervical esophagus was dissected. A protective sleeve (Ø35 mm) is installed inside the incision and was then covered by the matched protective cover (Figure 5). Three trocars (Ø5 mm) were placed into the cover, and CO2 insufflation is performed into the mediastinum with 10 mmHg to make artificial mediastinal emphysema (Figure 6). The assistant provides an excellent view by a 5-mm 30-degree lens through a trocar, while the surgeon inserted a Maryland forceps and an upper mediastinal retractor through the other two trocars. Firstly, along the left side of the esophagus, the upper mediastinal retractor has pushed the esophagus to the right.
The tissue around esophagus was coagulated and cut by the Maryland forceps. The blunt dissection of tissues was downward to the edge of the aortic arch. The thoracic duct was carefully protected. Secondly, the upper mediastinal retractor has lifted the esophagus, and the Maryland forceps was used along the anterior of spine downwards to make blunt dissection and cut the tissue around the esophagus to the azygos vein, which was until clearly showed. Thirdly, the upper mediastinal retractor has pulled the esophagus to the left. Along the right side of the groove between trachea and esophagus, the Maryland forceps was used downwards to make blunt dissection and cut off the tissue so that the peripheral lymph nodes were attached to the esophagus and removed in en bloc. The upper mediastinal retractor was lifted the trachea tissue at the lower margin of the left main bronchus, the lymph nodes behind carina of trachea were resected. During this procedure, the azygos vein and membranous tracheal wall should be completely protected. Because the origin of the left RLN lain at the lower margin of the aortic arch, blunt dissection was necessary to deal with the tissues between the esophagus and the left RLN. It was easy to mobilize the middle esophagus along with the spinal-esophageal space by blunt dissection. The lymph nodes around the left RLN was removed by endoscopic scissors, keeping the left RLN attached to the side of the trachea. The esophagus was mobilized as much as possible until the Maryland forceps could not further do.
If the preoperative imaging findings showed lymph node metastasis around the right RLN, lymph node dissection should be performed around the right RLN. Shortly, the upper mediastinal retractor has lifted the trachea near the thyroid. The right vagus nerve was found and exposed; the Maryland forceps was used along the right vagus nerve upwards to the right RLN while the right subclavian artery is a critical anatomical marker in surgery. Through the single-port left cervical incision, blunt dissection was performed in the lymph nodes around the right vagus nerve and the right RLN.
For laparoscopic operation, the incisions of the 5 ports for the laparoscopic operation were performed as described. One, the surgeon performed the procedure by making two 1 cm incisions at 3 cm from the umbilicus. Next, the assistant did a 1 cm incision under the right costal margin and a 5 mm incision under the xiphoid process. The photographer was placed through an incision about 3 cm below the left costal margin as the laparoscopic port (Figure 1B). The stomach was dissected by the conventional method, and the lymph nodes around the stomach were removed.
The right gastroepiploic artery was reserved and the short gastric artery, the ultrasonic scalpel was used to resect the gastrocolonic ligament and the gastrosplenic ligament. The gastric coronary vein and left gastric artery were exposed and ligated by Hemlock; by way of dissecting the surrounding lymph nodes. The ultrasonic scalpel was used to open the esophageal hiatus; two lower mediastinal retractors were placed to expose the esophageal hiatus. The Maryland forceps was used to dissect the tissues bluntly and removed lymph nodes near the esophageal hiatus and the lower esophagus. The perforation was not achieved until the lower esophagus was completely mobilized.
The esophagus was cut off at the neck, and a nail anvil was inserted into a pouch which was made in the proximal end, and then the purse stitching was tightened and fixed to prepare for anastomosis, while the distal esophagus was closed with suture. The esophagus and stomach were pulled out through a 5-cm subxiphoid incision. The linear stapling device was reconstructed for a 4 cm wide tubular stomach. The tubular stomach might be carried up to the neck through the esophageal bed or retrosternal approach. The circular stapler device anastomosed the tubular stomach and cervical esophagus. Gastrointestinal decompression was performed by inserting a gastric tube through the anastomosis. A nutrition tube was inserted and fixed in the jejunum through the subxiphoid incision, which was provided access to enteral nutrition. The mediastinal drainage tube was indwelling, and each incision was closed with appropriate sutures. It was the end of the operation.
Comment
Majority of the institutions perform MIE with the transthoracic approach (1). This is due to thoracic surgeons’ familiarity with the transthoracic approach and clear surgical view (11). Recent developments in endoscopic surgical devices and techniques have enabled underdone transmediastinal approach with total pneumomediastinum assistance for esophageal cancer (9); we developed a novel method for single-port inflatable mediastinoscopy and laparoscopic surgery for esophageal cancer. It solves the problems of conventional transmediastinal approach with a limited surgical view, insufficient mediastinal lymphadenectomy, single lung ventilation, and cardiopulmonary complications (10). Upper mediastinal lymphadenectomy by a transmediastinal approach depends on the anatomy of the RLN and the esophagus; the lymph nodes along the RLN can be dissected easily under endoscopy vision by transmediastinal approach, especially those around the aortic arch and the lymph nodes behind carina of trachea. The mediastinum around the aortic arch and left tracheobronchial angle are critical in lymphadenectomy along the left RLN in this area. A pneumomediastinum not only expands the mediastinal space but also makes it easy to identify these structures including bronchial arteries, vagus nerves, lymphatic vessels and mechanical retraction of the aortic arch. The right visual field provided by the transmediastinal approach is helpful and safe to the surgeon. It saves a lot of time that upper mediastinal operation and laparoscopic operation are undergone simultaneously, if two groups of surgeons stand like we demonstrated. It is a very good surgical technique that is worth to promote.
Acknowledgments
Funding: This study was supported by National Key R&D Program of China (2018YFC0910603).
Footnote
Provenance and Peer Review: This article was commissioned by the Editorial Office, Mediastinum for the series “Mediastinoscopic Surgery” published in Mediastinum. The article has undergone external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/med.2019.05.01). The series “Mediastinoscopic Surgery” was commissioned by the editorial office without any funding or sponsorship. QC serves as the unpaid Guest Editor of this series. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and national research committees and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Wu PC, Posner MC, Wu PC, et al. The role of surgery in the management of oesophageal cancer. Lancet Oncol 2003;4:481-8. [Crossref] [PubMed]
- Niclauss N, Chevallay M, Frossard JL, et al. Surgical strategy for early stage carcinoma of the esophagus. Chirurg 2018;89:339-46. [Crossref] [PubMed]
- Klink CD, Binnebösel M, Otto J, et al. Intrathoracic versus cervical anastomosis after resection of esophageal cancer: a matched pair analysis of 72 patients in a single center study. World J Surg Oncol 2012;10:159. [Crossref] [PubMed]
- Kutup A, Nentwich MF, Bollschweiler E, et al. What should be the gold standard for the surgical component in the treatment of locally advanced esophageal cancer: transthoracic versus transhiatal esophagectomy. Ann Surg 2014;260:1016-22. [Crossref] [PubMed]
- Takahashi C, Shridhar R, Huston J, et al. Esophagectomy from then to now. J Gastrointest Oncol 2018;9:903-9. [Crossref] [PubMed]
- Achim F, Constantinoiu S. Recent Advances in Minimally Invasive Esophagectomy. Chirurgia (Bucur) 2018;113:19-37. [Crossref] [PubMed]
- Seto Y, Mori K, Aikou S. Robotic surgery for esophageal cancer: Merits and demerits. Ann Gastroenterol Surg 2017;1:193-8. [Crossref] [PubMed]
- Fujiwara H, Shiozaki A, Konishi H, et al. Transmediastinal approach for esophageal cancer: A new trend toward radical surgery. Asian J Endosc Surg 2019;12:30-6. [Crossref] [PubMed]
- Fujiwara H, Shiozaki A, Konishi H, et al. Single-Port Mediastinoscopic Lymphadenectomy Along the Left Recurrent Laryngeal Nerve. Ann Thorac Surg 2015;100:1115-7. [Crossref] [PubMed]
- Wang X, Li X, Cheng H, et al. Single-Port Inflatable Mediastinoscopy Combined With Laparoscopic-Assisted Small Incision Surgery for Radical Esophagectomy Is an Effective and Safe Treatment for Esophageal Cancer. J Gastrointest Surg 2019; [Epub ahead of print]. [Crossref] [PubMed]
- Gisbertz SS, Hagens E, Ruurda JP, et al. The evolution of surgical approach for esophageal cancer. Ann N Y Acad Sci 2018;1434:149-55. [Crossref] [PubMed]
Cite this article as: Wang X, Li X, Huo W, Cheng H, Zhang B, Zhong H, Wang R, Cao Q. The procedure of single-port inflatable mediastinoscopy and laparoscopic surgery for radical esophagectomy. Mediastinum 2019;3:22.